ISSN: 2688-562X
Case Report
Second Line Chemotherapy with Carboplatin-Vinorelbine after first line Paclitaxel-Capecitabine for Recurrent or Metastatic Squamous Cell Head and Neck Carcinoma
Kenneth Jensen1*, Emma Victoria Sjölund Schmidt1, Niels Gyldenkerne2, and Jens Bentzen3
- 1Department of Oncology, Aarhus University Hospital, Denmark
- 2Department of Oncology, Odense University Hospital, Denmark
- 3Department of Oncology, Herlev University Hospital, Denmark
- *Corresponding author: Kenneth Jensen, Danish Center of Particle Therapy, Department of Oncology, Aarhus University Hospital, Palle Juul Jensens Boulevard 25. 8200 Aarhus N, Denmark, Tel: 45 21284108;
- Received: Jul 11, 2019 Accepted: Jul 23, 2019 Published: Jul 25, 2019
Abstract
Background
Recurrent squamous cell head and neck cancer has a grave prognosis. Available systemic therapies have limited efficiency and significant toxicity. At our institutions, many patients receive first line chemotherapy with paclitaxel and capecitabine, as the most effective 3-drug regimen, with cisplatin, 5-FU and cetuximab often leads to significant toxicity.
Objective
We wanted to retrospectively assess the efficiency of our preferred 2nd line treatment after paclitaxel and capecitabine: carboplatin and vinorelbine.
Design
Retrospective analysis
Setting
At 3 academic tertiary centers, we identified 87 patients, who have had paclitaxel and capecitabine as first line treatment and carboplatin and vinorelbine as second line treatment for recurrent/ metastatic squamous cell head and neck cancer.
Measurements
Data on treatment response, side effects and survival
Results
The median number of chemotherapy series was 3 and toxicity was pronounced with 63% experiencing grade ≥ 3 side effects including 33% febrile neutropenia and 1 toxic death. Side effects were more pronounced with intravenous infusion than oral capsules of vinorelbine; overall grade ≥ 3 toxicity 73% (iv) vs. 24% () (p ≤ 0,001). Only 12% had an objective response to therapy, and any improved symptom was only noted for 18% of the patients. Median overall survival was 163 (95% CI; 120-206) days.
Conclusion
Toxicity was pronounced and response rated very limited in this series of second line chemotherapy. Treatment with monoclonal antibodies, especially against PD-L1, but also EGFR, seems more attractive, and no anti-neoplastic treatment might be a better choice.
Keywords:
Squamous Cell Head and Neck Cancer; Recurrent; Metastatic; Chemotherapy; Second line
Introduction
Background
Recurrent squamous cell head and neck cancer (SCCHNC) carries a very grave prognosis. There is limited efficiency of systemic treatment, with a median survival of 10 months with the most efficient treatment available, which combines Cisplatin, 5-FU and cetuximab [1]. The 3-drug regimen has severe side effects, and often patients with recurrent head and neck cancer have co-morbidity, high age or psychosocial problems that make the three-drug combination less appealing [2]. Also many patients receive concomitant cisplatin during curative radiotherapy, raising the possibility of resistance [3].Therefore,other drug combinations are often used, and in Denmark there is widespread use of paclitaxel-capecitabine as first line chemotherapy for patients in less than optimal general condition, due to comparable response and superior toxicity profile [4,5], despite no data from a direct comparison. The results of second line chemotherapy are even more bleak with poor efficacy and very little evidence to guide the choice of therapy as previous therapy is often very heterogeneous in available reports [6]. Nevertheless, immunotherapy has now become an accepted second line therapy due to both superior survival and toxicity profile, compared to chemotherapy or anti epidermal growth factor (EGFR) antibodies [7-9]. Unfortunately, more than 80% of patients do not respond to immunotherapy or EGFR targeted therapy [7-9]. The symptoms of uncontrolled, often loco-regionally progressive, head and neck cancer are so severe that treatment attempts may be justified, in selected patients, even with conventional chemotherapy, in spite of the some side effects of treatment.
Objective
Due to the limited efficiency of established treatments we have there for analyzed our results with carboplatin-vinorelbine ,our preferred second line regimen after paclitaxe-capecitabine, in order to guide treatment for those patients who are not candidate for, or progress after, immunotherapy.
Methods
Three academic tertiary referral centers(Aarhus, Odense and Herlev University Hospital), treating approximately 60% of all Danish head and neck cancer patients, participated in the study as they shared the general guidelines of using paclitaxel-capecitabine for most patients as first line treatment, and carboplatin-vinorelbine as the preferred second line treatment. Patients were identified in the DAHANCA (Danish Head and Neck Cancer Group) database or in institutional databases of administered chemotherapy. The data were collected retrospectively from all available sources: the DAHANCA database, medical records and treatment forms. The data collection and analysis were approved by the Danish Health and Medicines Authority. Patients began 2nd line chemotherapy between March 2007 until February 2015.
Chemotherapy was administered as carboplatin intravenous (iv), AUC 5 at 3 weeks interval. Oral vinorelbine was administered as 60 mg/m2 day 1 and 8 of the first series and dose escalated to 80 mg/m2 day 1 and 8 for the remaining series in case of absence of neutropenia or other significant toxicity. In case of dysphagia and before the oral formulation (per os: po) was introduced, vinorelbine was administered as 30 mg/m2 iv. The patients were evaluated after 3 series, if the treatment was not stopped, due to clinical progression or side effects.
Survival analysis was reported using Kaplan-Meyer analysis and covariates for survival were analyzed using Cox regression analysis. Toxicity was graded according to CTC AE 4.0.
Results
The vast majority (79%) have had radiotherapy with curative intent as their initial treatment. Most patients had primary pharynx cancer. Over all 77% patients had stage ≥ 3 and 7% had metastatic disease at time of initial diagnosis. The median number of first line chemotherapy series was 6 (Table 1).
Table 1: Baseline characteristics. Patients with recurrent/ metastatic SCCHNC
N=87 |
|
Men (%) |
72 |
Primary Tumor Site (%) |
11 (13) |
Age at first Carboplatin-Vinorelbine Mean, (Range) |
62 (24-79) |
P16 (pos/neg/unknown) (n=87) (%) |
17*/34/36 (20/39/41) |
Primary disease Stage (n=84) (%) |
10 (12) |
Smoking (n=87) |
13 (15) |
Previous RT (Unknown; None; Palliative; Curative) (%) |
2; 13;6;79 |
Concomitant cisplatin (%) |
16 (18) |
First Line Paclitaxel-Capecitabine |
|
Number of series (Median; Mean; Range) |
6; 6,4;1-27 |
Dose Reduction (%) |
47 |
*10 patients with p16+ oropharynx cancer
Median number of 2nd line chemotherapy series was 3, 46% were reduced in dose and 63% experienced some kind of grade ≥ 3 toxicity, mainly hematological, including one toxic death from febrile neutropenia (Table 2). Only half the patients stopped treatment after three, six or nine series, indicating that clinical progression or prohibitive side effects between evaluations were common. Administration of vinorelbine intravenously was predictive of neutropenia (p=0.005), infection (p<0.001), anemia (p=0.24), sensory neuropathy (p=0.03), injection site reaction (p=0.03) and overall grade ≥ 3 toxicity (73% (iv) vs. 24% ()) (p ≤ 0.001). The risk of febrile neutropenia (37% (iv) vs 18% (po)) did not reach statistical significance (Kendall tau c-test).
Table 2: Treatment. Second line Carboplatin-Vinorelbine after Paclitaxel- Capecitabine for recurrent/ metastatic SCCHNC
N=87 |
|
|
Administration (i.v/p.o.) (%) |
70/17 (80/20) |
|
Number of series (Median; Mean; Range) |
3; 3.2; 1-9 |
|
Dose Reduction (%) |
46 |
|
Side Effects |
Grade 0; 1; 2; 3; 4; 5 (%) |
% ≥ Grade 3 |
Neutropenia |
43; 3;6;16;32;0 |
48 |
Febrile Neutropenia |
67;-;-;31;1;1 |
33 |
Infection |
70; 0; 2; 23; 5; 0 |
28 |
Thrombocytopenia |
86; 7; 1; 2; 3; 0 |
5 |
Anemia |
53; 2; 35;10; 0; 0 |
10 |
Mucositis |
97; 1; 0; 2; 0; 0 |
2 |
Peripheral sensory neuropathy |
94; 0; 3; 2; 0; 0 |
2 |
Peripheral motor neuropathy |
98; 0; 1; 1; 0; 0 |
1 |
Injection site reaction (at risk (iv vino) n=70) |
93; 4; 3; 0; 0; 0 |
0 |
Diarrhea |
98; 0; 1; 1; 0; 0 |
1 |
Obstipation |
97; 1; 0; 2; 0; 0 (2) |
2 |
Nausea |
97; 0; 2; 1; 0; 0 (1) |
1 |
Vomiting |
97; 0; 2; 1; 0; 0 (1) |
1 |
Any Grade ≥3 (%) |
63 |
|
Response (NE;PD;NC;PR) (%) |
2;59; 28; 12 |
|
Subjective response, any (Yes; No)* (%) |
18; 82 |
|
*From the medical record, e.g. decreased dyspnea, improved general condition and decrease in local discomfort at tumor site.
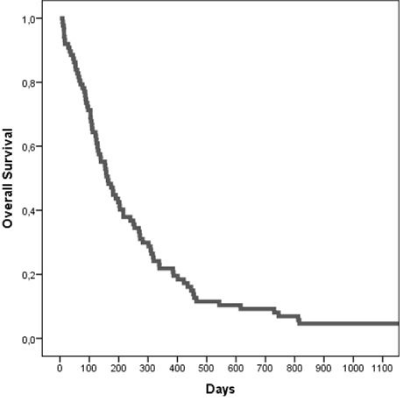
Response rates was very limited, with no complete responses, 12% partial responses (PR) and 28% stable disease (SD) as best response, making progressive disease (PD) the most likely “best” treatment response. Any kind of positive subjective response was stated in the medical records for 18% of the patients. Median overall survival was 163 (95% CI; 120-206) days (Figure 1). Survival was lower among patients with PD compared to other (Hazard 3.72 (95% CI: 2.29- 6.06). Age, gender and time since previous chemotherapy series did not predict for any side effects nor survival (data not shown).
Discussion
The median number of series of first line chemotherapy was in line with previous publications [4]. Only half the patients stopped 2nd line treatment after formal evaluation (at three, six or nine series), stressing that the patients and treatment per se should be evaluated continuously. Toxicity, including hematological grade 3 toxicity was somewhat higher compared to first line carboplatin-vinorelbine, often used in Denmark for lung cancer [10], and an older report on carboplatin-vinorelbine for first line treatment of recurrent head and neck cancer [11]. Even though paclitaxel-capecitabine does not have significant hematological toxicity, it may impact the reserve capacity of the bone marrow. Deteriorated general condition and progressive cancer may be an alternative explanation for the high risk of hematological toxicity. There were no differences in survival or chance of response between the oral and iv administration of vinorelbine, but more side effects, especially hematological, were seen using iv administration. This difference in the risk of toxicity is in contrast with results in breast cancer [12], but in line with results from the treatment of lung cancer [10,13]. In the present population, any increase in toxicity there might be, could have been enhanced by the fact that many patients had iv vinorelbine because of swallowing difficulties, i.e. more symptomatic, than those who could swallow capsules.
Response and overall survival were in line with some recently published studies [14,15], and overview [16], on chemotherapy for platinum resistant patients, but inferior to reports with the combination of paclitaxel plus an antibody [17], or tyrosin kinase inhibitor [18], which were not a relevant option for our patients. We are currently conducting a randomized study examining the addition of cetuximab to out standard first line chemotherapy of paclitaxel and capecitabine.
The drawback of this study was the retrospective design. Responses as well as side effects were extracted from medical record, meaning that response might not be in line with the RECIST criteria. Side effects, especially grade 1-2 nausea and vomiting, may be under reported.
The advantage of the present study is the homogeneity of the first line treatment, making it very useful for evaluating carboplatinvinorelbine in this specific setting, as well as providing information for the treatment of other patients for whom cisplatin was not an option for first line treatment. Our data on survival are complete, with no patients lost to follow up.
Conclusion
Toxicity was pronounced and response rated very limited in this series of second line chemotherapy. Intravenous and oral administration of vinorelbine did not seem equivalent, with significantly more toxicity with iv. vinorelbine. Treatment with monoclonal antibodies, especially against programmed death ligand 1 (PD-L1), but also EGFR, seems more attractive, and no anti neoplastic treatment at all, might probably lead to a better quality of life and comparable survival. Further research on new drugs and new therapeutic strategies are necessary.
References
- Vermorken JB, Mesia R, Rivera F, Remenar E, Kawecki A, Rottey S, et al. Platinum- Based Chemotherapy plus Cetuximab in Head and Neck Cancer. N Engl J Med. 2008; 359: 1116-1127.
- Fisher MD, Fernandes AW, Olufade TO, Miller PJ, Walker MS, Fenton M. Patient Characteristics and Costs in Recurrent or Refractory Head and Neck Cancer: Retrospective Analysis of a Community Oncology Database. Clin Ther. 2018; 40: 562-573.
- Bentzen J, Toustrup K, Eriksen JG, Primdahl H, Andersen LJ, Overgaard J. Locally advanced head and neck cancer treated with accelerated radiotherapy, the hypoxic modifier nimorazole and weekly cisplatin. Results from the DAHANCA 18 phase II study. Acta Oncol. 2015; 54: 1001-1007.
- Bentzen JKD, Kristensen CA, Overgaard M, Rytter C, Jensen K, Hansen HS. A non platinum regimen for the treatment of recurrent or metastatic squamous cell carcinoma of the head and neck region. Results from an extended phase II study with paclitaxel and capecitabine. Front Oncol. 2018; 8: 243.
- Bentzen JD, Hansen HS. Phase II analysis of paclitaxel and capecitabine in the treatment of recurrent or disseminated squamous cell carcinoma of the head and neck region. Head Neck. 2007; 29: 47-51.
- Lala M, Chirovsky D, Cheng JD, Mayawala K. Clinical outcomes with therapies for previously treated recurrent / metastatic head-and-neck squamous cell carcinoma (R / M HNSCC ): A systematic literature review. Oral Oncol. 2019; 84: 108-120.
- Chow LQM, Haddad R, Gupta S, Mahipal A, Mehra R, Tahara M, et al. Antitumor activity of pembrolizumab in biomarker-unselected patients with recurrent and/ or metastatic head and neck squamous cell carcinoma: Results from the phase Ib KEYNOTE-012 expansion cohort. J Clin Oncol. 2016; 34: 3838-3845.
- Ferris RL, Blumenschein G, Fayette J, Guigay J, Colevas AD, Licitra L, et al. Nivolumab for Recurrent Squamous-Cell Carcinoma of the Head and Neck. N Engl J Med. 2016; 375: 1856-1867.
- Vermorken JB, Trigo J, Hitt R, Koralewski P, Diaz-Rubio E, Rolland F, et al. Openlabel, uncontrolled, multicenter phase II study to evaluate the efficacy and toxicity of cetuximab as a single agent in patients with recurrent and/or metastatic squamous cell carcinoma of the head and neck who failed to respond to platinum-based therapy. J Clin Oncol. 2007; 25: 2171-2177.
- Jensen LH, Osterlind K, Rytter C. Randomized cross-over study of patient preference for oral or intravenous vinorelbine in combination with carboplatin in the treatment of advanced NSCLC. Lung Cancer. 2008; 62: 85-91.
- Kornek GV, Scheithauer W, Glaser C, Toth J, Duman M, Formanek M, et al. Vinorelbine and carboplatin in recurrent and/or metastatic squamous cell carcinoma of the head and neck. Oncology. 1999; 56: 24-27.
- Lorusso V, Cinieri S, Giampaglia M, Ciccarese M, Tinelli A, Chiuri V, et al. Intravenous versus oral vinorelbine plus capecitabine as second-line treatment in advanced breast cancer patients. A retrospective comparison of two consecutive phase II studies. Breast. 2010; 19: 214-218.
- Hirsh V, Desjardins P, Needles BM, Rigas JR, Jahanzeb M, Nguyen L, et al. Oral versus intravenous administration of vinorelbine as a single agent for the first-line treatment of metastatic nonsmall cell lung carcinoma (NSCLC): A randomized phase II trial. Am J Clin Oncol Cancer Clin Trials. 2007; 30: 245-251.
- Siano M, Infante G, Resteghini C, Cau MC, Alfieri S, Bergamini C, et al. Outcome of recurrent and metastatic head and neck squamous cell cancer patients after first line platinum and cetuximab therapy. Oral Oncol. 2017; 69: 33-37.
- Machiels JPH, Haddad RI, Fayette J, Licitra LF, Tahara M, Vermorken JB, et al. Afatinib versus methotrexate as second-line treatment in patients with recurrent or metastatic squamous-cell carcinoma of the head and neck progressing on or after platinum-based therapy (LUX-Head & Neck 1): An open-label, randomised phase 3 trial. Lancet Oncol. 2015; 16: 583-594.
- de Andrade DA, Machiels JP. Treatment options for patients with recurrent or metastatic squamous cell carcinoma of the head and neck, who progress after platinum-based chemotherapy. Curr Opin Oncol. 2012; 24: 211-217.
- Sosa AE, Grau JJ, Feliz L, Pereira V, Alcaraz D, Muñoz-García C, et al. Outcome of patients treated with palliative weekly Paclitaxel plus Cetuximab in recurrent head and neck cancer after failure of platinum-based therapy. Eur Arch Oto-Rhino- Laryngology. 2014; 271: 373-378.
- Soulières D, Faivre S, Mesía R, Remenár É, Li SH, Karpenko A, et al. Buparlisib and paclitaxel in patients with platinum-pretreated recurrent or metastatic squamous cell carcinoma of the head and neck (BERIL-1): a randomised, double-blind, placebocontrolled phase 2 trial. Lancet Oncol. 2017; 18: 323-335.
Citation: Kenneth Jensen. Second Line Chemotherapy with Carboplatin-Vinorelbine after first line Paclitaxel-Capecitabine for Recurrent or Metastatic Squamous Cell Head and Neck Carcinoma. Clin Oncol. 2019; 2(2): 1012.
PDF DownloadClinics Oncology © All Rights Reserved. 2018